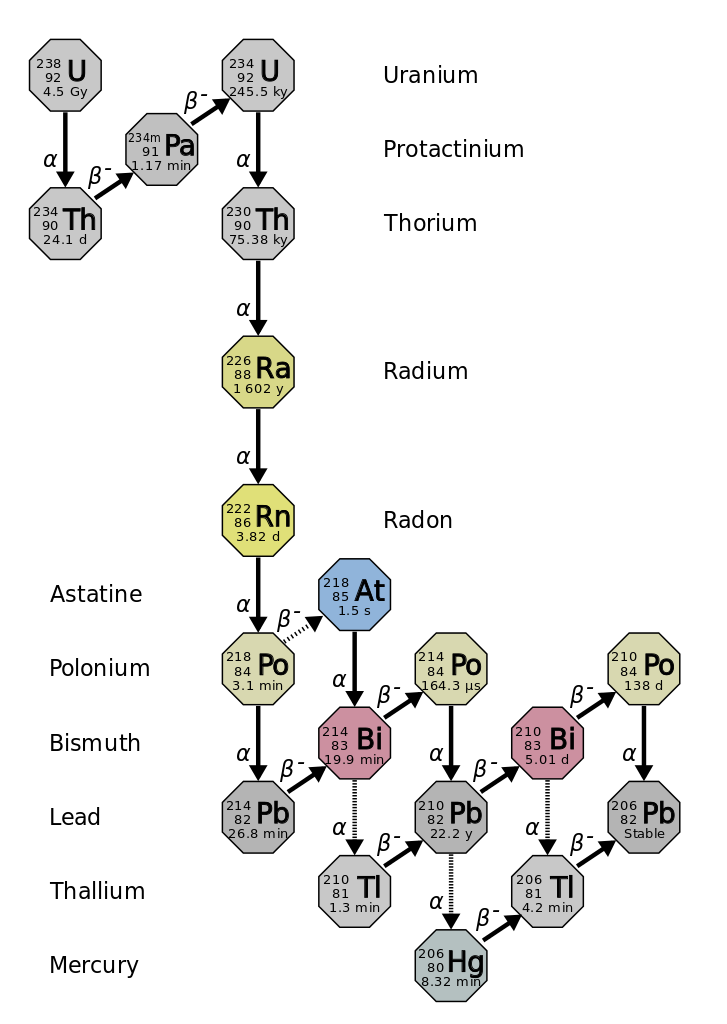
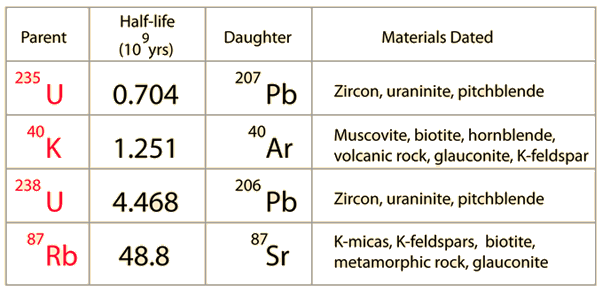
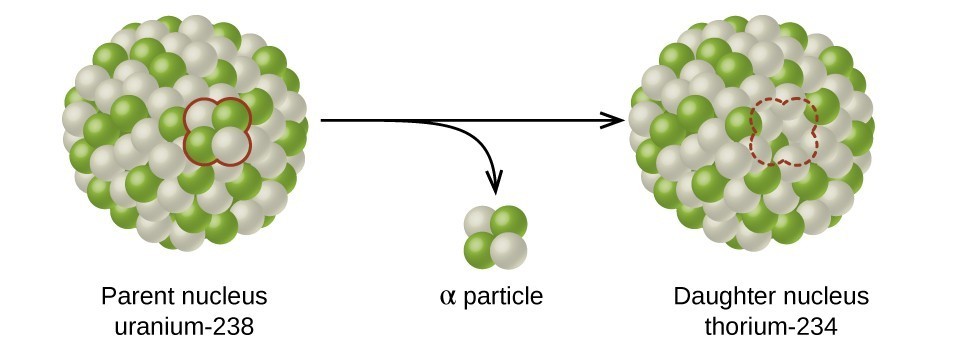
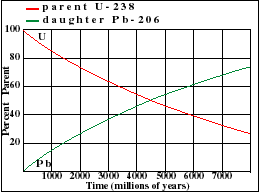
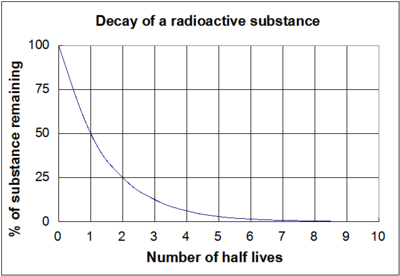
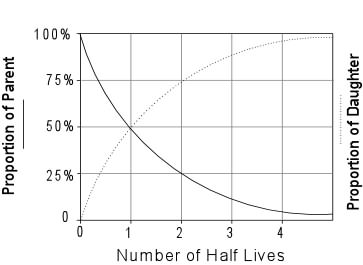
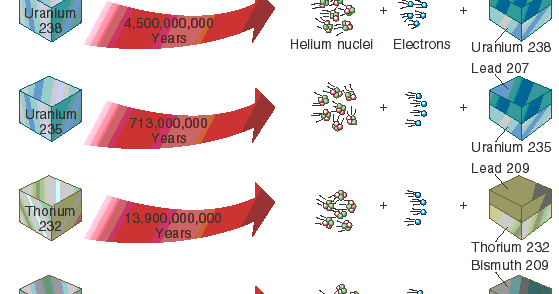
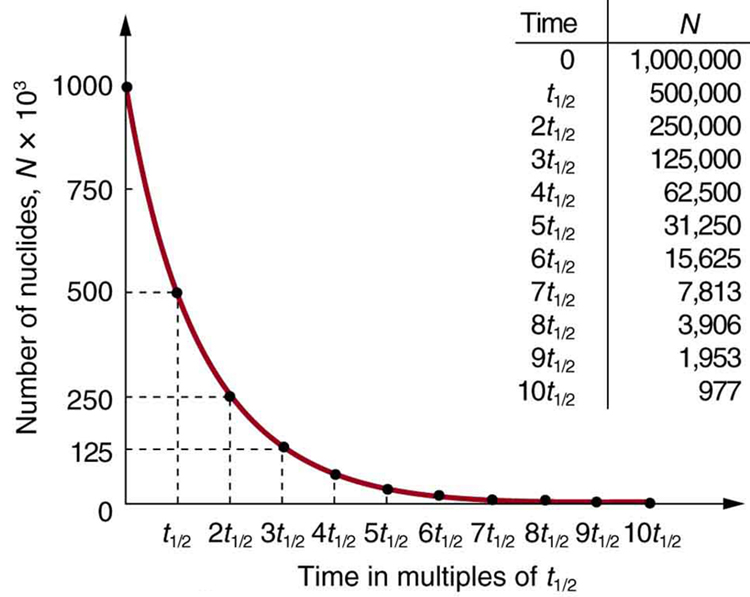
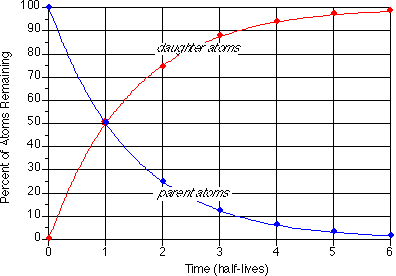
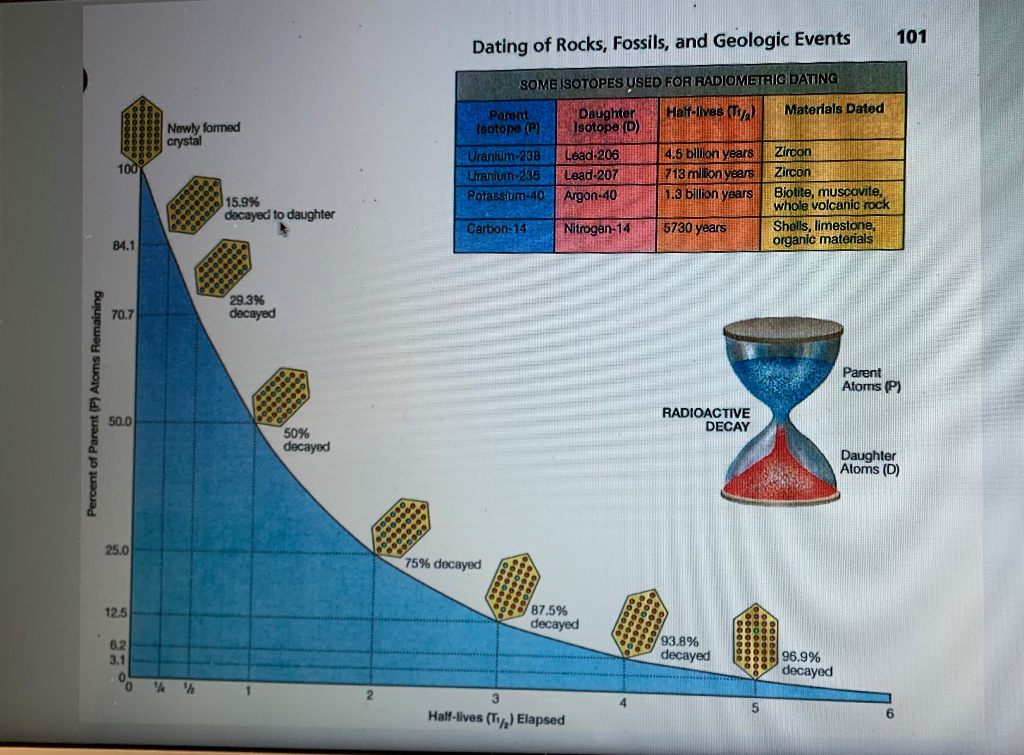
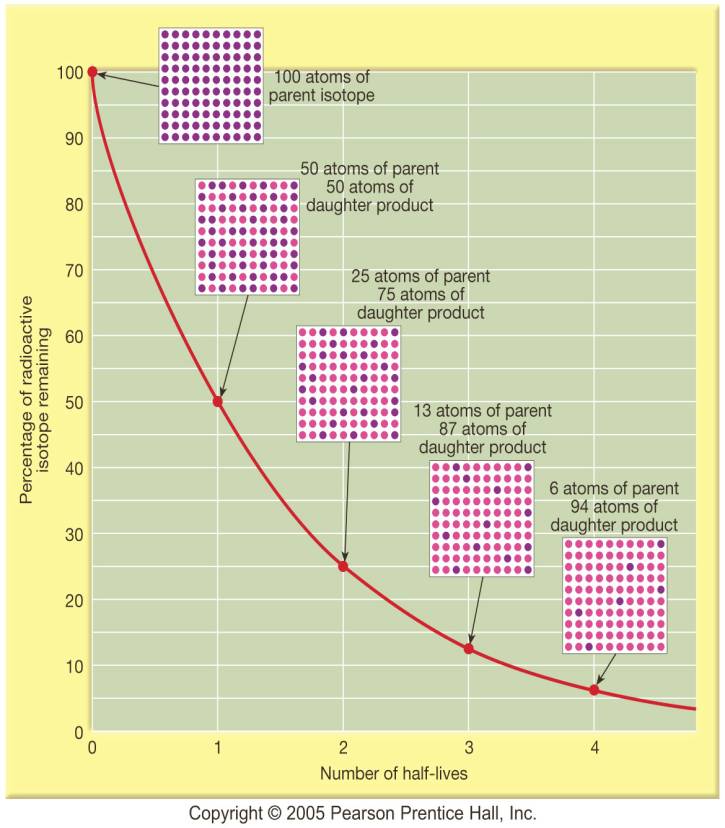
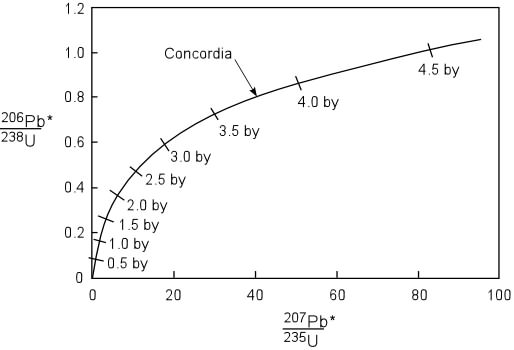
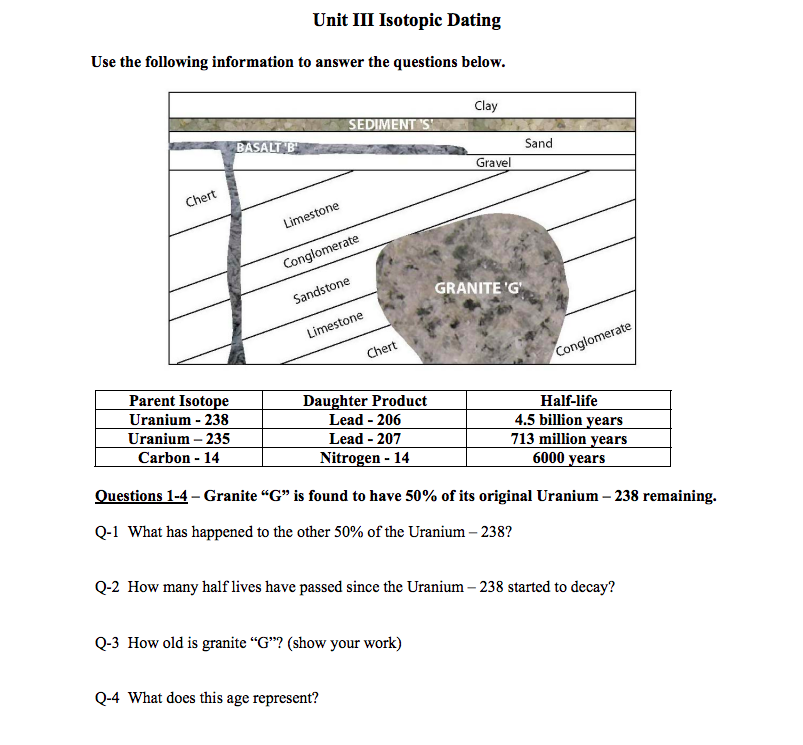
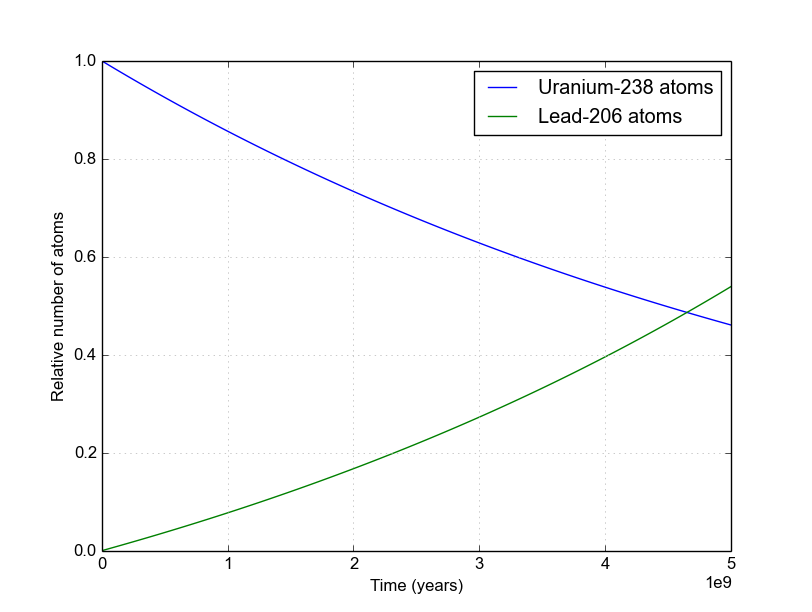
A rock is found to contain U-238 and Pb-206 in the ratio of 3 : 2. If `t\"\"_(1//2)` of U-238 is `4. - YouTube

SOLVED:The most common isotope of uranium, ^238 U, has atomic mass 238.050783 u. Calculate (a) the mass defect; (b) the binding energy (in MeV); (c) the binding energy per nucleon.

21 - chemistry - 21: Radioactive Decay Recognize common modes of radioactive decay Identify common - Studocu

The final product of U^(238) is Pb^(206). A sample of pitchblende contains 0.0453 g of Pb^(206) for every gram of U^(238) present in it. Supposing that the mineral pitchblende formed at the
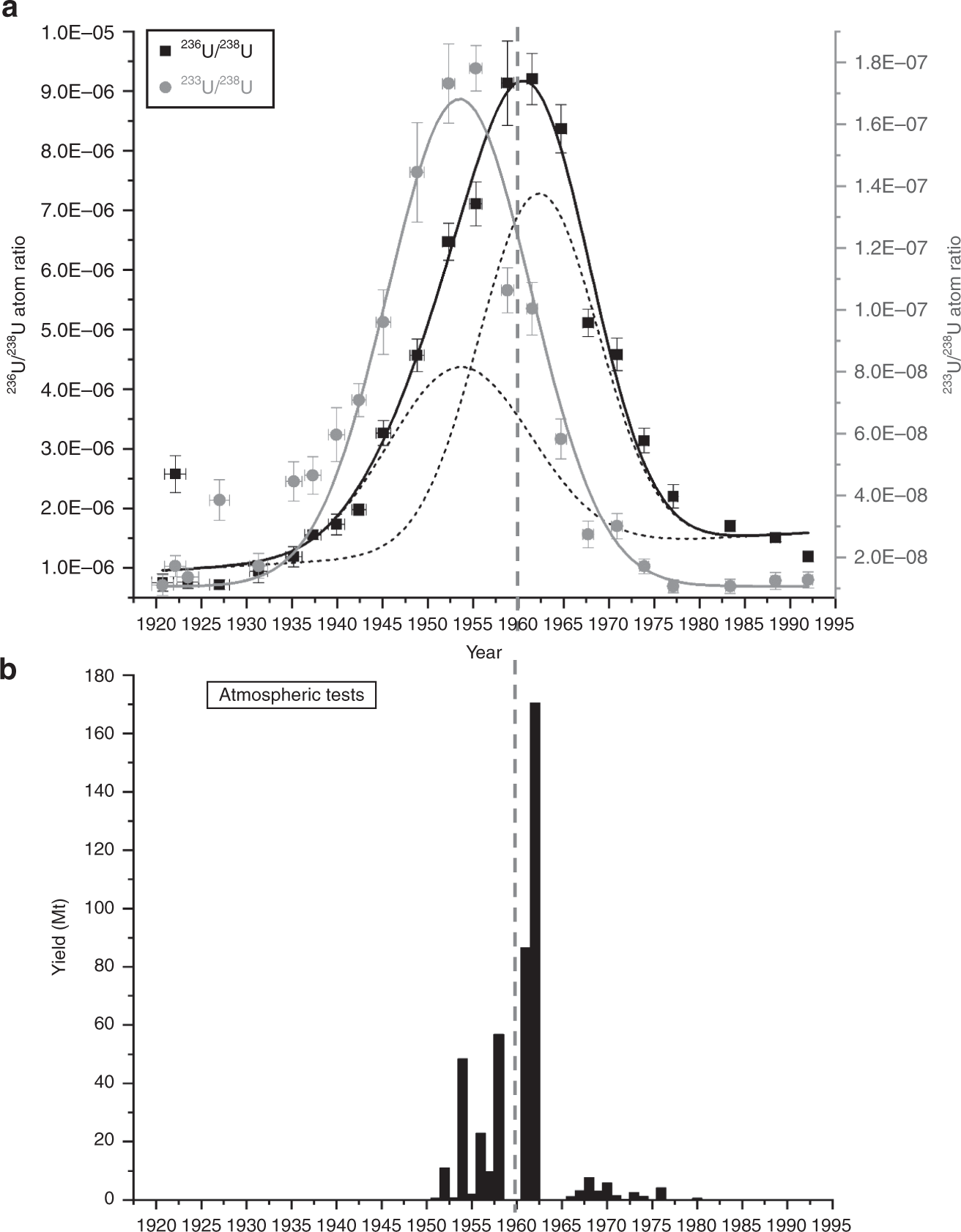
233U/236U signature allows to distinguish environmental emissions of civil nuclear industry from weapons fallout | Nature Communications