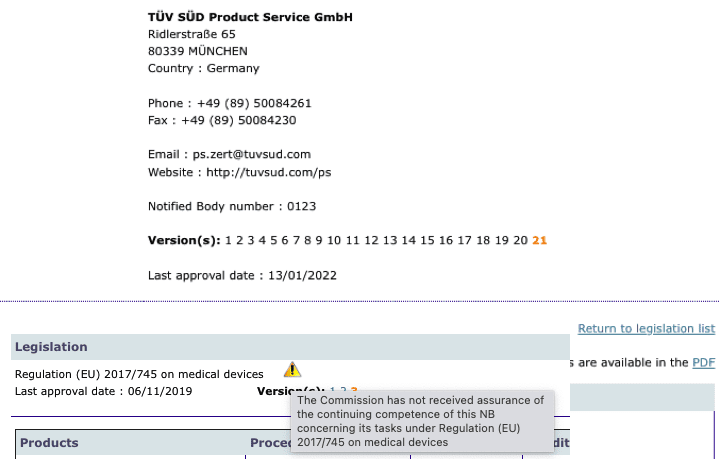
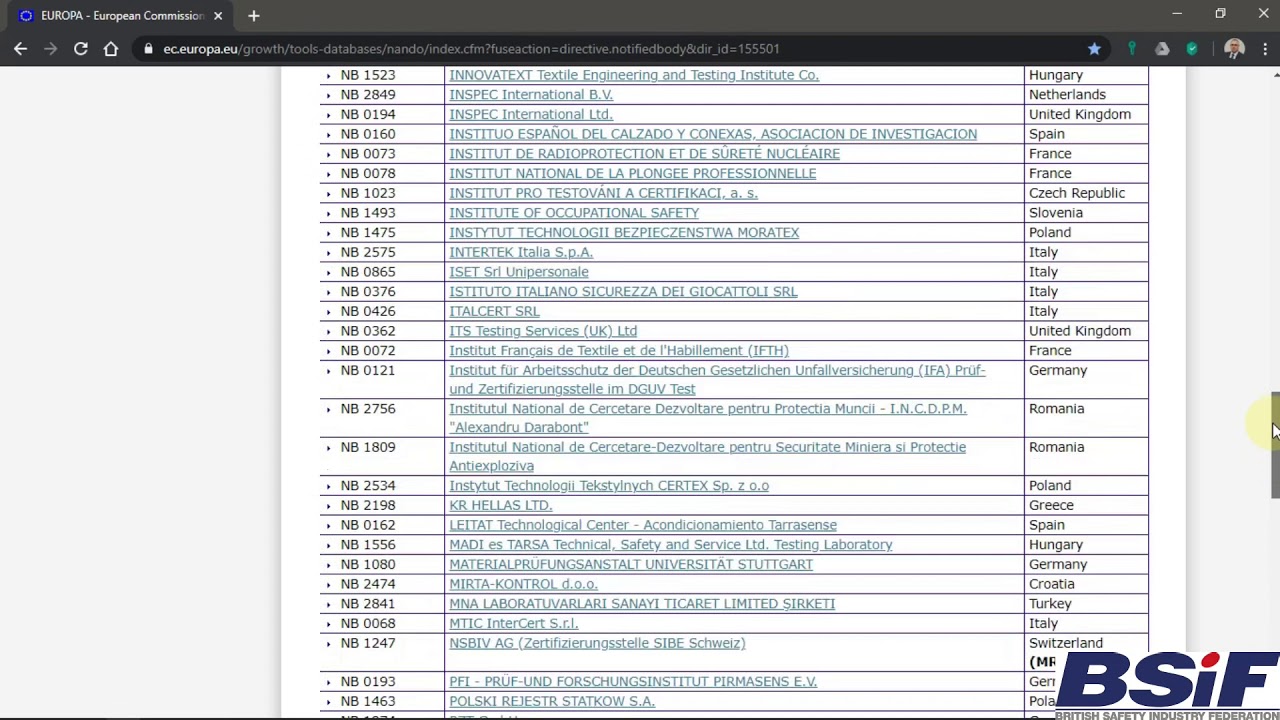
MDR: 29 Notified Bodies on NANDO · MDlaw – Information platform on European medical device regulations

MDR: 26 Notified Bodies on NANDO & Swiss economic operator's requirements updated! · MDlaw – Information platform on European medical device regulations
![Glossary] EU MDR and IVDR: The core terms you should know - Pharma Serialization, Aggregation and Track and Trace Software by SoftGroup Glossary] EU MDR and IVDR: The core terms you should know - Pharma Serialization, Aggregation and Track and Trace Software by SoftGroup](https://www.softgroup.eu/wp-content/uploads/2020/10/Untitled-design-38-1280x720.png)