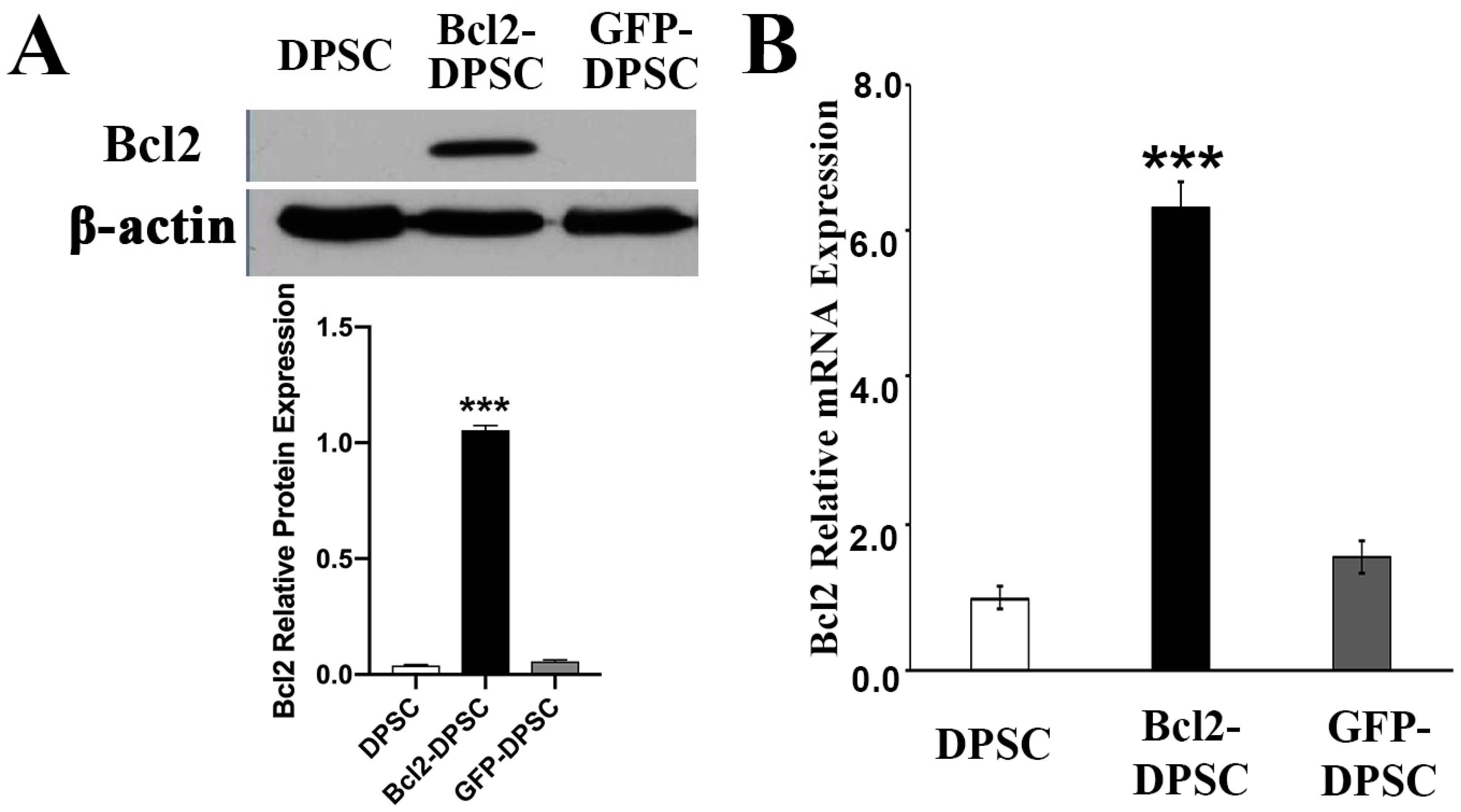
IJMS | Free Full-Text | Bcl-2 Overexpression and Hypoxia Synergistically Enhance Angiogenic Properties of Dental Pulp Stem Cells

IJMS | Free Full-Text | Recombinant Human Arresten and Canstatin Inhibit Angiogenic Behaviors of HUVECs via Inhibiting the PI3K/Akt Signaling Pathway

Human Umbilical Vein Endothelial Cells Survive on the Ischemic TCA Cycle under Lethal Ischemic Conditions | Journal of Proteome Research
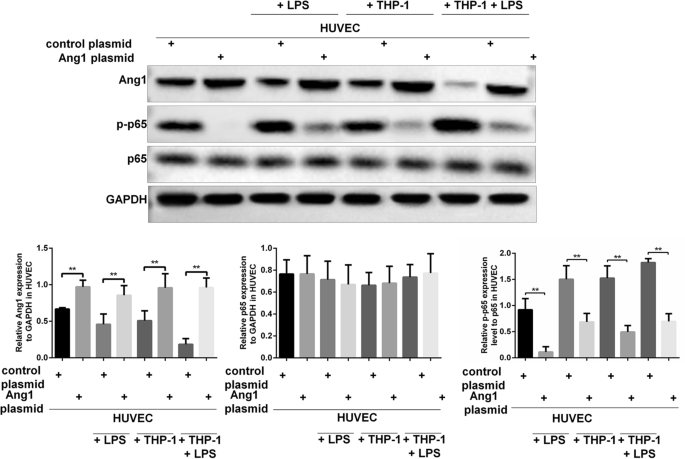
Macrophages enhance lipopolysaccharide induced apoptosis via Ang1 and NF-κB pathways in human umbilical vein endothelial cells | Scientific Reports
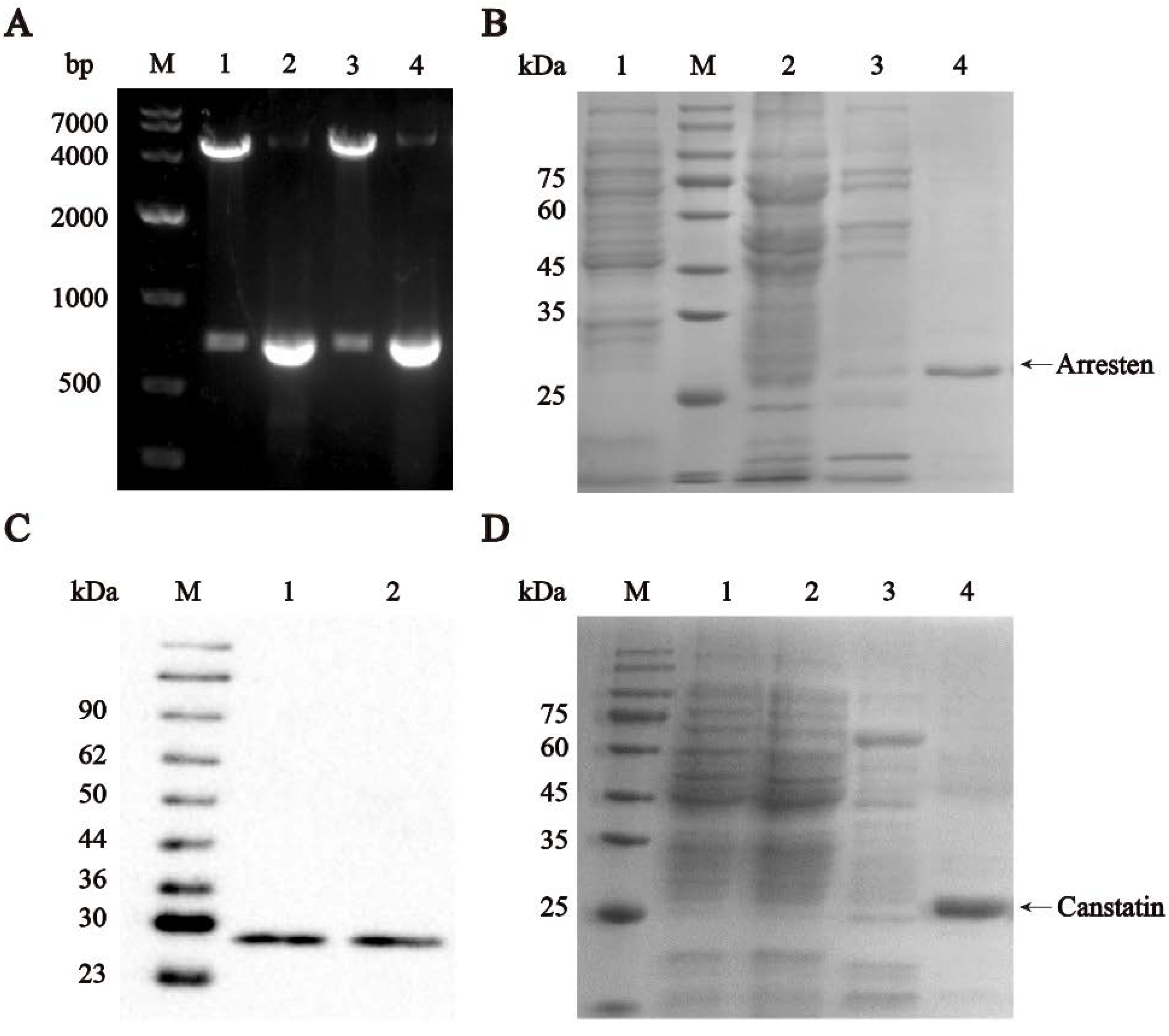
IJMS | Free Full-Text | Recombinant Human Arresten and Canstatin Inhibit Angiogenic Behaviors of HUVECs via Inhibiting the PI3K/Akt Signaling Pathway

Silencing of OIP5-AS1 Protects Endothelial Cells From ox-LDL-Triggered Injury by Regulating KLF5 Expression via Sponging miR-135a-5p - Frontiers

Effects of PDE4i on the HUVEC cytotoxicity and proliferation. (a and b)... | Download Scientific Diagram
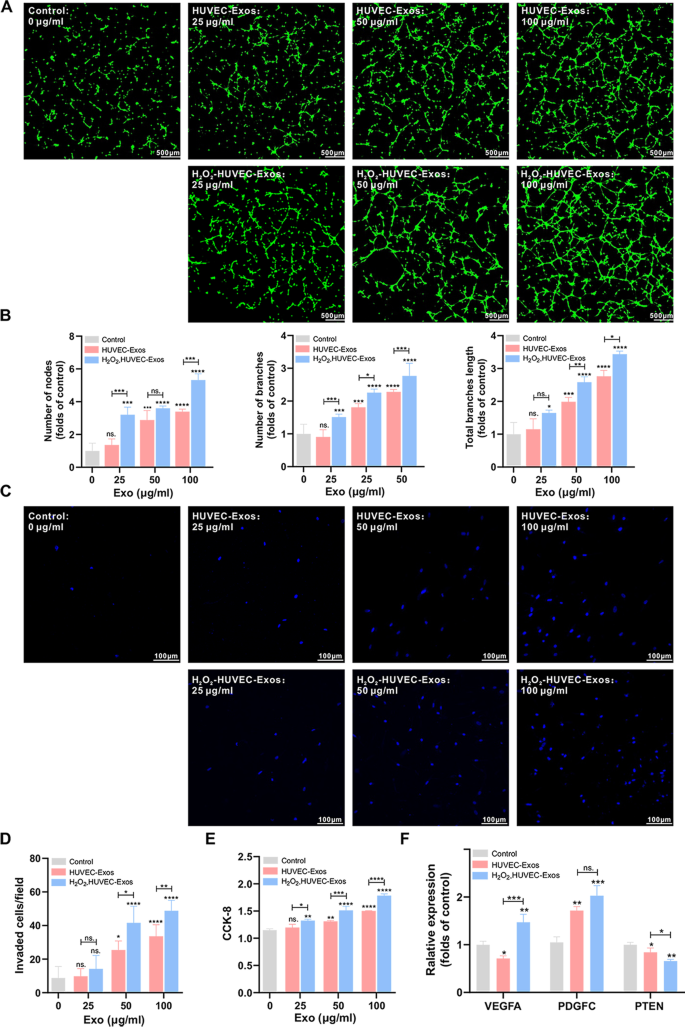
Oxidative stress-induced endothelial cells-derived exosomes accelerate skin flap survival through Lnc NEAT1-mediated promotion of endothelial progenitor cell function | Stem Cell Research & Therapy | Full Text

Astaxanthin suppresses End MT by LOX-1 pathway in ox-LDL-induced HUVECs - Zhongsheng Zhu, Jinyu Li, Rui Tong, Xiaorong Zhang, Bo Yu, 2022
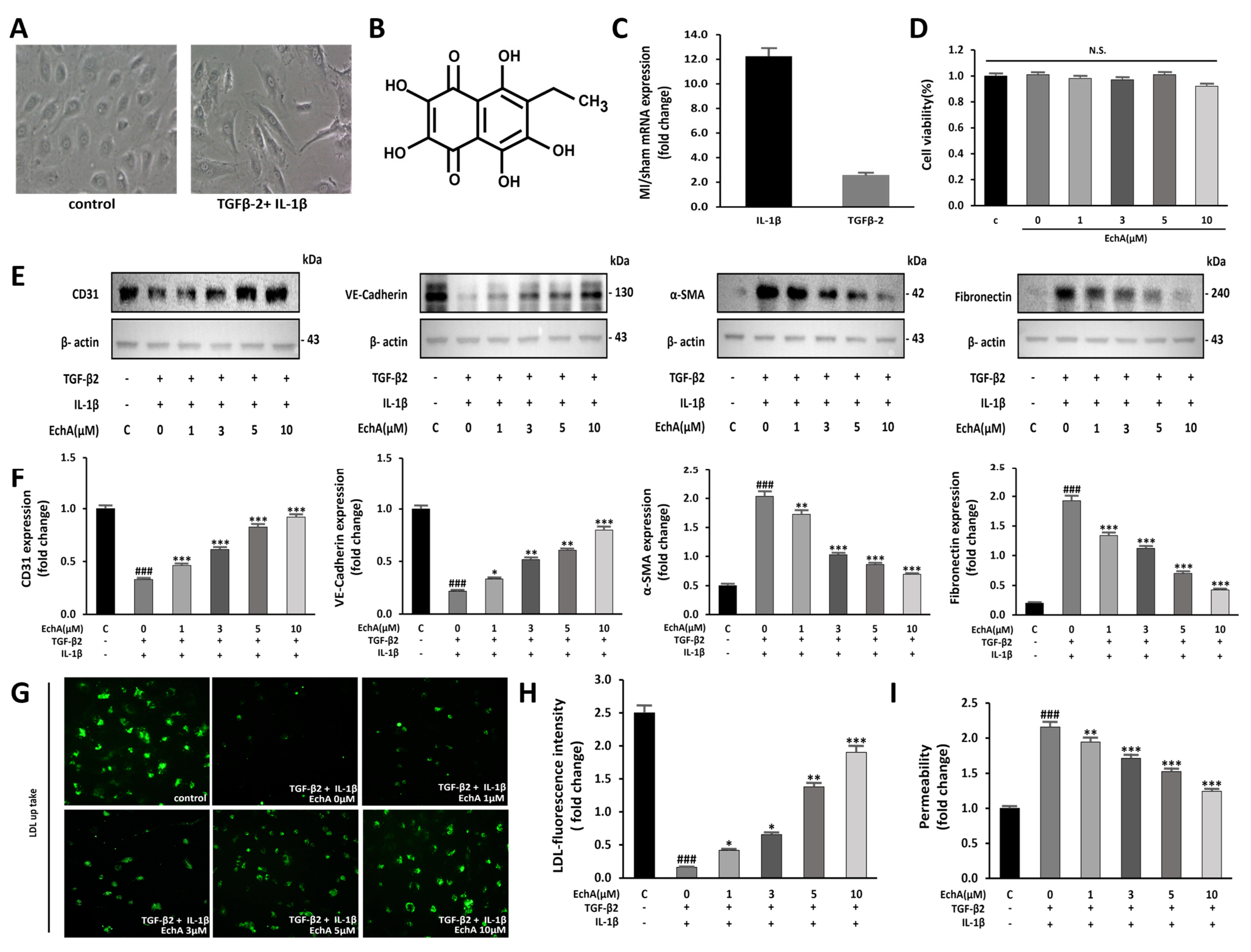
Marine Drugs | Free Full-Text | Regulation of Inflammation-Mediated Endothelial to Mesenchymal Transition with Echinochrome a for Improving Myocardial Dysfunction

13-Methylberberine improves endothelial dysfunction by inhibiting NLRP3 inflammasome activation via autophagy induction in human umbilical vein endothelial cells | Chinese Medicine | Full Text
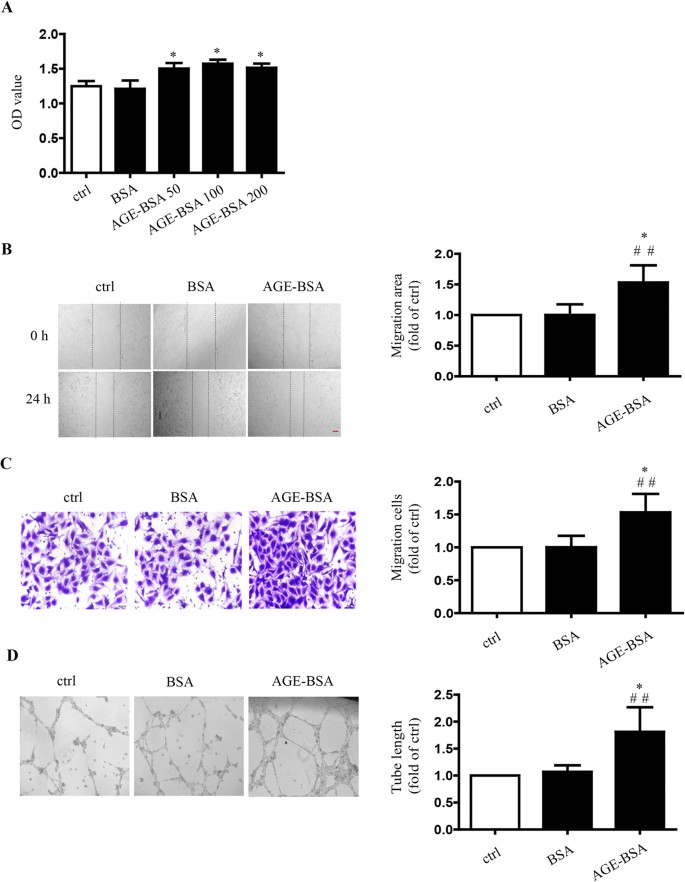
Role of Moesin in Advanced Glycation End Products-Induced Angiogenesis of Human Umbilical Vein Endothelial Cells | Scientific Reports
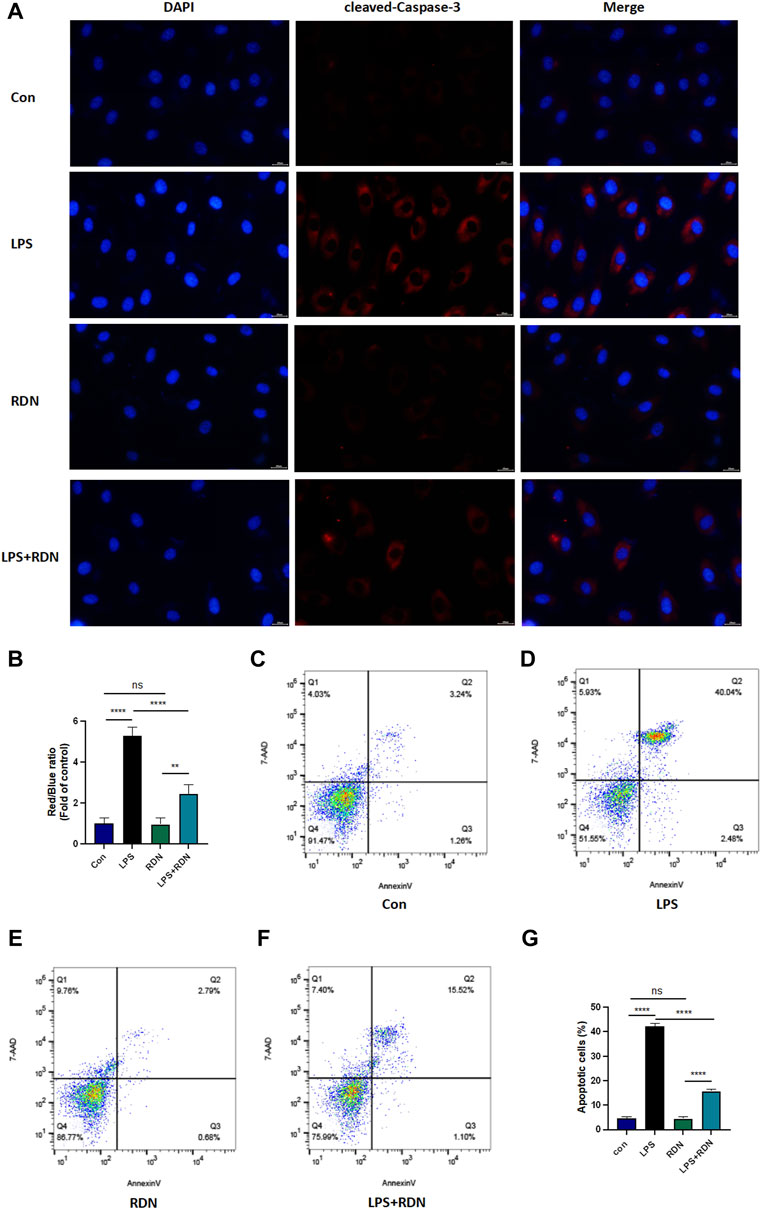
Frontiers | Reduning Attenuates LPS-Induced Human Unmilical Vein Endothelial Cells (HUVECs) Apoptosis Through PI3K-AKT Signaling Pathway
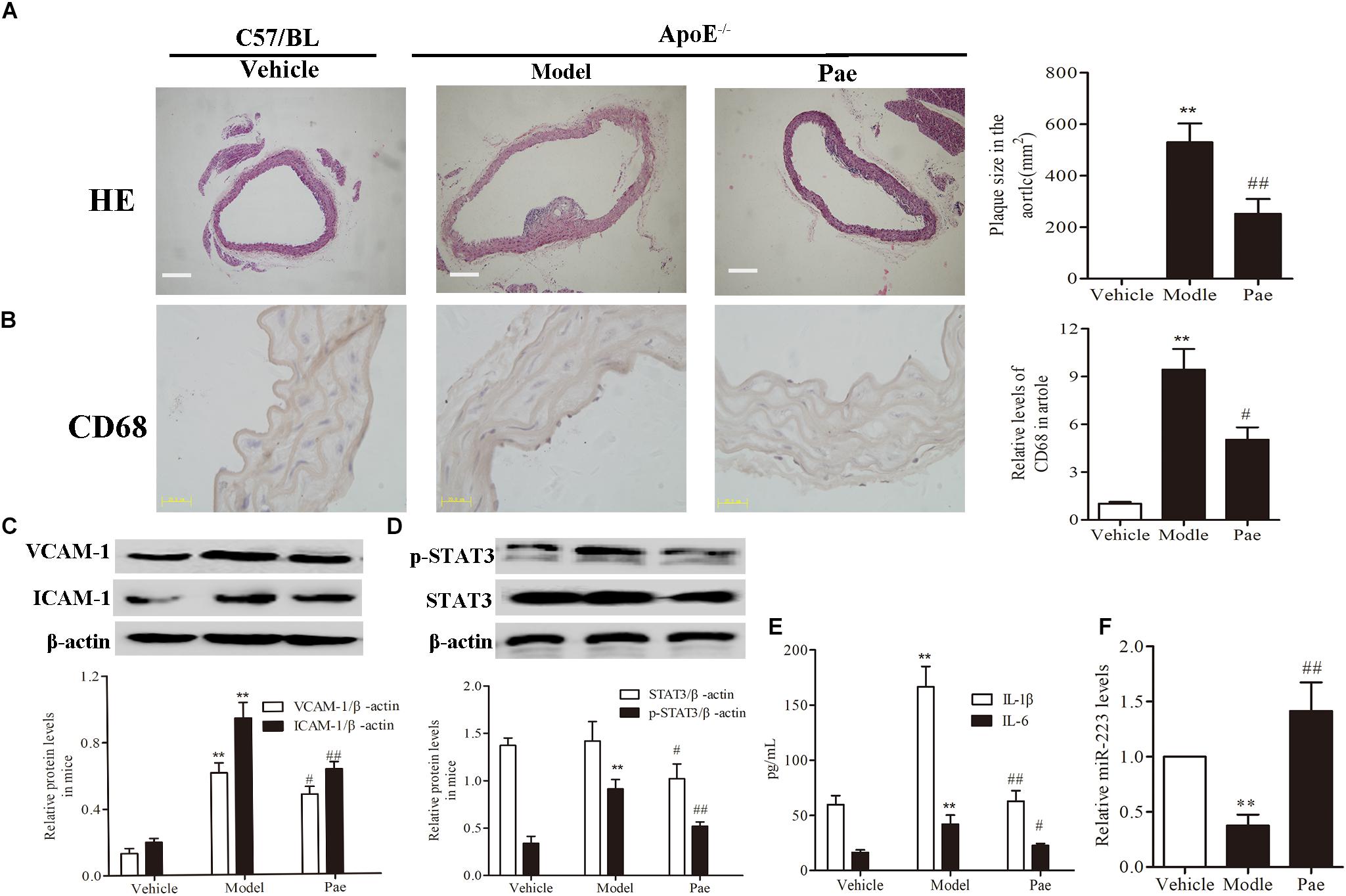
Frontiers | Paeonol Attenuated Inflammatory Response of Endothelial Cells via Stimulating Monocytes-Derived Exosomal MicroRNA-223
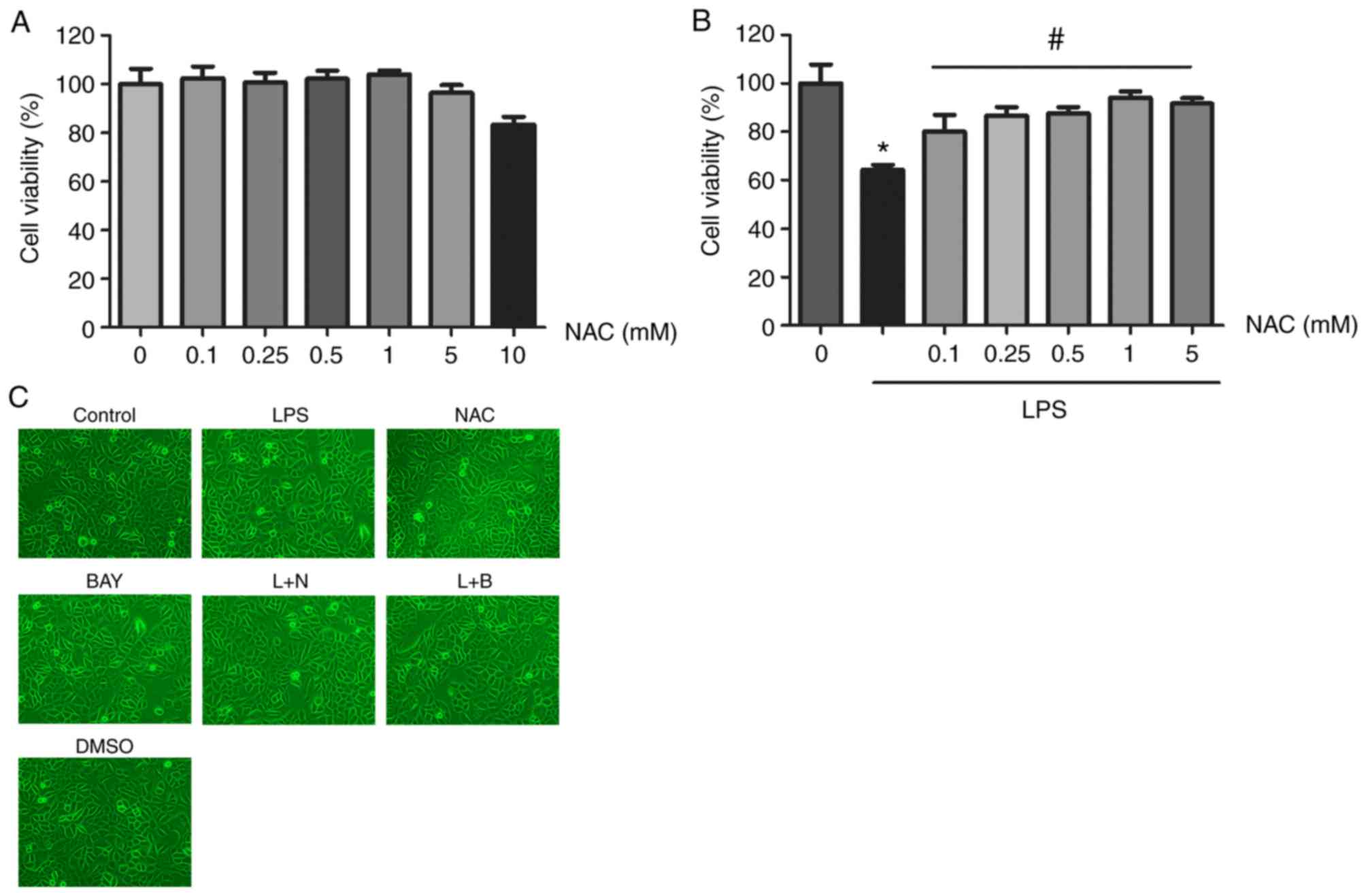
N‑acetyl cysteine protects HUVECs against lipopolysaccharide‑mediated inflammatory reaction by blocking the NF‑κB signaling pathway

Myricetin ameliorates ox-LDL-induced HUVECs apoptosis and inflammation via lncRNA GAS5 upregulating the expression of miR-29a-3p | Scientific Reports
Circular RNA circVEGFC accelerates high glucose-induced vascular endothelial cells apoptosis through miR-338-3p/HIF-1α/VEGFA axis | Aging
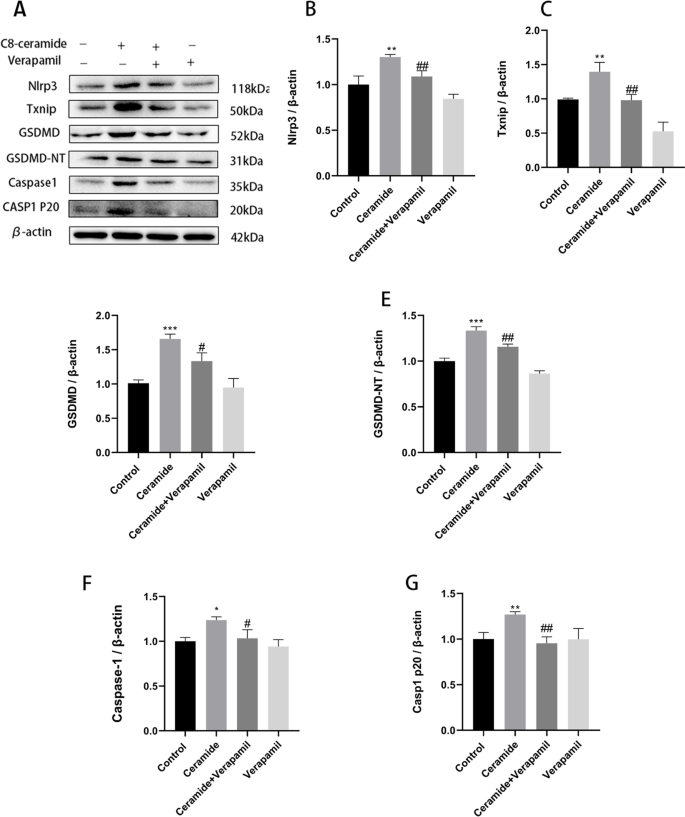
Ceramide induces pyroptosis through TXNIP/NLRP3/GSDMD pathway in HUVECs | BMC Molecular and Cell Biology | Full Text

MiR-410 inhibition induces HUVECs proliferation and represses ox-LDL-triggered apoptosis through activating STAT3 - ScienceDirect

Rapamycin inhibits ox-LDL-induced inflammation in human endothelial cells in vitro by inhibiting the mTORC2/PKC/c-Fos pathway | Acta Pharmacologica Sinica


