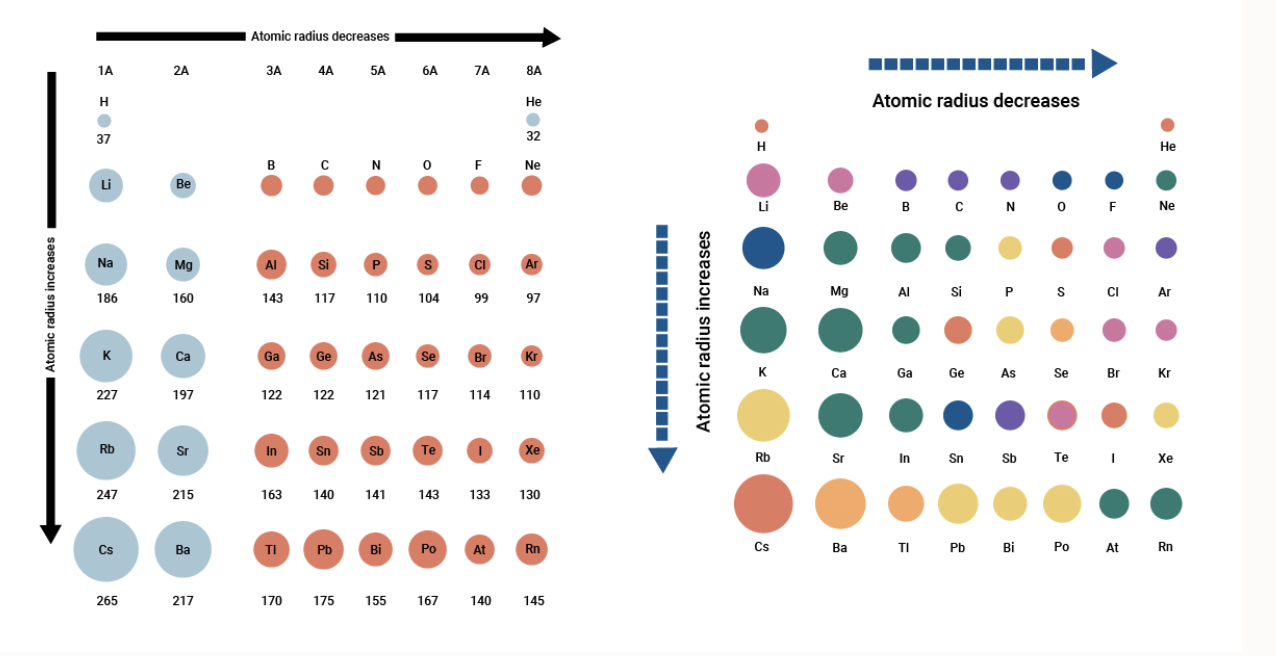
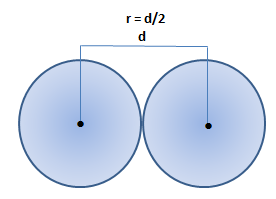
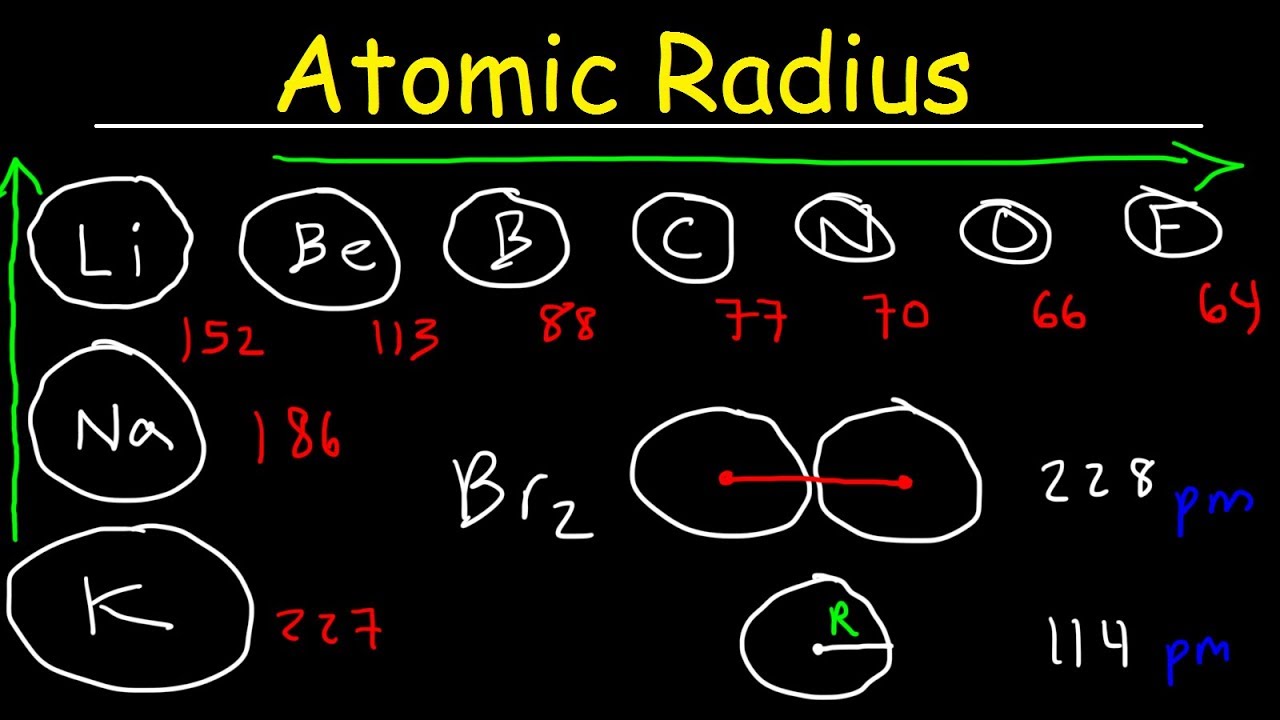
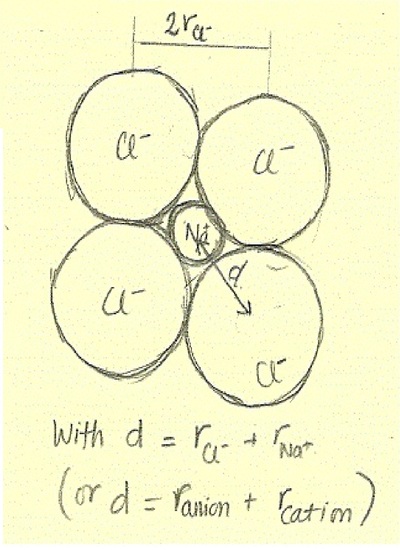What is the formula to calculate the radius of an orbit of the atom and velocity of the specific shell of the atom.

Calculate the radius of second Bohr orbit in hydrogen atom from the given data.Mass of electron = 9.1 × 10^-31Kg Charge on the electron = 1.6 × 10^-19C Planck's constant = 6.63 ×

Niobium has a density of 8.57 g/cm3 and crystallizes with the body-centered cubic unit cell. Calculate the radius of a niobium atom - Chemistry Stack Exchange